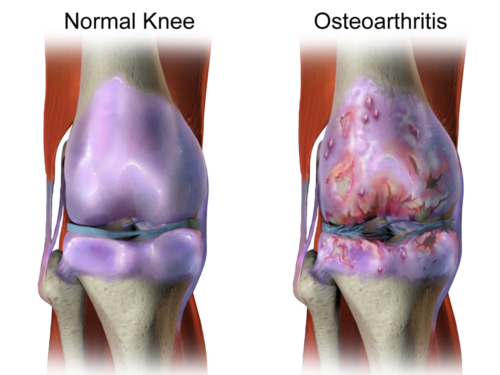
Some friends recently asked me about getting stem cell injections for painful knees with osteoarthritis (Figure 1). Should they push their doctors for this treatment? Were they, and consequently their stem cells, going to be too old if they waited?
I started my investigation at PubMed, searching for “stem cells” and “osteoarthritis,” which produced 1249 articles. Limiting the results to those in humans brought the number to 761, of which a little more than half were published since 2013 and in English. Despite adding the filter for human, the titles of the articles showed many of the results were fro
m animal studies. So, I added filters for clinical trial and review, and I added the word “knee” to the search. This yielded 38 articles of which 16 were original articles and the rest were reviews (Figure 2).
My take-home message from this research is that injection of autologous stem cells (those from the patient him- or herself, not from another source) is reasonably safe, but there is no conclusive evidence that it is effective or more effective than other options, such as injections with molecules or compounds that provide joint lubrication. In large part, this is because these studies are still relatively new, and many were designed to test safety with improvement in mobility and pain and reduction in tissue damage secondary outcomes of the studies. The studies were fairly short term; most followed the patients for 2 years or less. Many did not have very many subjects, and some did not have a randomized control design. Some lacked control subjects. Therefore, conclusions cannot be easily and confidently made, and the results cannot be confidently extended to the population. This does not mean that stem cell therapy may not eventually be highly effective, just that we do not yet have sufficient evidence for it, and we have not determined the optimal process for this type of treatment.
Stem cell injections in the knee are safe but effectiveness is not yet clearly established.
For people interested in pursuing stem cell therapy, I suggest joining a clinical trial. In this way, you can contribute to the scientific knowledge about the effectiveness of stem cell-based therapy for osteoarthritis of the knee. So far, the data are insufficient for FDA approval of the stem cell-based treatment for osteoarthritis. I predict that there will be optimal ways of pretreating stem cells to maximize their proper differentiation for restoring joint tissue and that there will be concomitant procedures (like surgically smoothing the cartilage or inducing microfractures or the use of artificial lubricants or hydrogel cushions) that may improve the response. More clinical trials are needed to find the best ways to leverage stem cell-based therapies for treating osteoarthritis.
Join a clinical trial…contribute to the knowledge about the effectiveness of stem cell-based therapy for osteoarthritis.
There are multiple therapeutic options for treating osteoarthritis, such as microsurfacing of the cartilage, injections of joint-lubricating compounds, nonsteroidal anti-inflammatory agents, and physical therapy. Only one FDA-approved therapy for osteoarthritis is a biologic agent, and it uses cells. MACI is a treatment that uses cultured chondrocytes (the cells that make cartilage) grown on a supporting material made from collagen from pigs. The chondrocytes are obtained from a biopsy from the patient’s knee, then grown on the collagen support membrane, and this cell-coated membrane is transplanted into the knee to replace damaged articular cartilage.
Stay tuned for upcoming articles about joint biology and the current and potential treatment options for arthritis.
Related Reading (Freely Available)
M. M. Richards, J. S. Maxwell, L. Weng, M. G. Angelos, J. Golzarian, Intra-articular treatment of knee arthritis: from anti-inflammatories to products of regenerative medicine. Phys. Sportsmed. 44, 101-108 (2016). PubMed
J. Freitag, D. Bates, R. Boyd, K. Shah, A. Barnard, L. Huguenin, A. Tenen, Mesenchymal stem cell therapy in the treatment of osteoarthritis: reparative pathways, safety, and efficacy- a review. BMC Musculoskelet. Disord. 17, 230 (2016). PubMed
P. K. Gupta, A. Chullikana, M.Rengasmy, N. Shetty, V. Pandey, V. Agarwal, S. Y. Wagh, P. K. Vellotare, D. Damodaran, P. Viswanathan, C. Thei, S. Balasubramanian, A. S. Majumdar, Efficacy and safety of adult human bone marrow-derived, cultured, pooled, allogenic mesenchymal stromal cells (Stempeucel): preclinical and clinical trial in osteoarthritis of the knee joint. Arthritis Res. Ther. 18, 301 (2016). PubMed
J. M. Lamo-Espinosa, G. Mora, J. F. Blanco, F. Granero-Moltó, J. M. Nuñez-Córdoba, C. Sánchez-Echenique, J. M. Bondía, J. Dámaso Aquerreta, E. J. Andreu, E. Ornilla, E. M. Villarón, A. Valentí-Azcárate, F. Sánchez-Guijo, M. Consuelo del Cañizo, J. Ramón Valentí-Nin, F. Prósper, Intra-articular injection of two different doses of autologous bone marrow mesenchymal stem cells versus hyaluronic acid in the treatment of knee osteoarthritis: multicenter randomized controlled clinical trial (phase I/II). J. Transl. Med. 14, 246 (2016). PubMed
Related Reading (Fee)
D. Saris, A. Price, W. Widuchowski, M. Bertrand-Marchand, J. Caron, J. O. Drogset, P Emans, A. Podskubka, A. Tsuchida, S. Kili, D. Levine, M. Brittberg, SUMMIT Study Group. Matrix-applied characterized autologous cultured chondrocytes versus microfracture: Two-year follow-up of a prospective randomized trial. Am. J. Sports Med. 42, 1384-1394 (2014).PubMed
Clinical Trials
Clinical trials for osteoarthritis, knee, and cell: Search results
Related Resources
Knee Anatomy Video (accessed on 5 January 2018)
Knee Osteoarthritis Video (accessed on 5 January 2018)
FDA warns about stem cell therapies. U.S. Department of Health and Human Services, FDA. (accessed on 5 January 2018) https://www.fda.gov/ForConsumers/ConsumerUpdates/ucm286155.htm
MACI. U.S. Department of Health and Human Services, FDA. (accessed on 5 January 2018) https://www.fda.gov/BiologicsBloodVaccines/CellularGeneTherapyProducts/ApprovedProducts/ucm533177.htm
MACI. Vericel Corporation (accessed on 5 January 2018) http://www.maci.com/
Cite as: N. R. Gough, Stem Cell Injections for Osteoarthritic Knees. BioSerendipity (8 January 2018). https://www.bioserendipity.com/2018/01/08/stem-cells-for-osteoarthritic-knees/