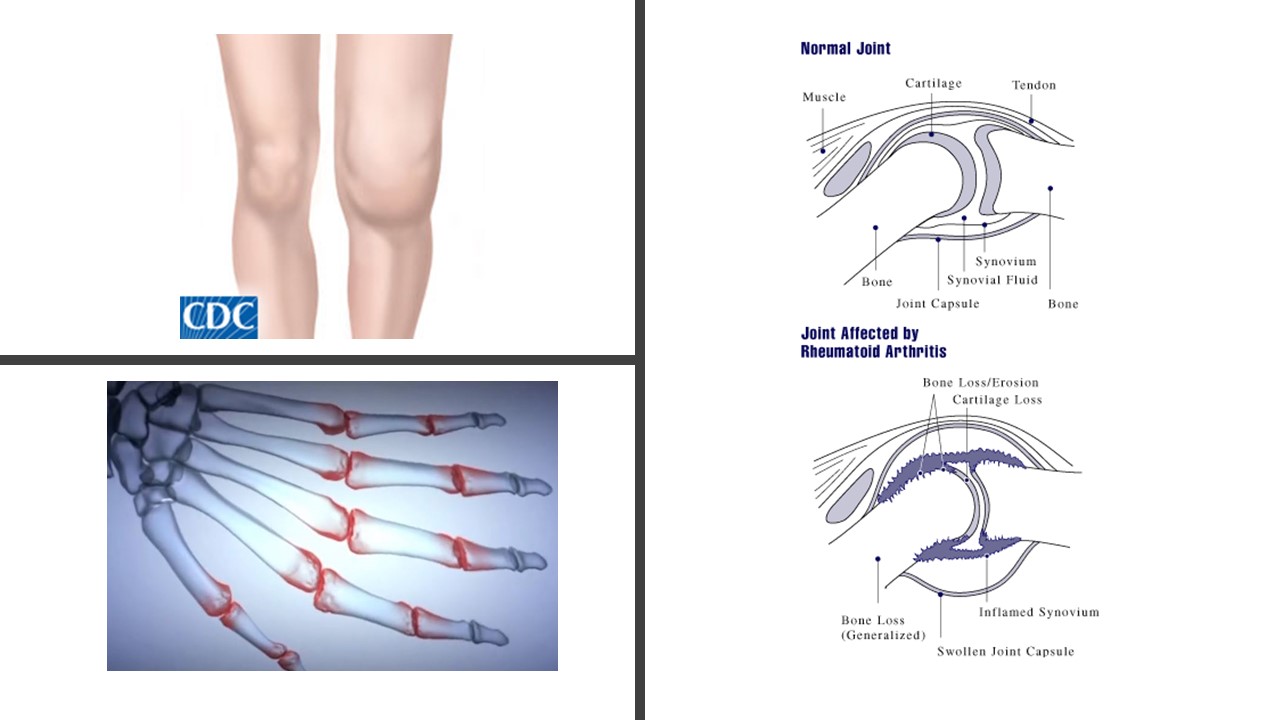
Unlike osteoarthritis, rheumatoid arthritis is an autoimmune disease (1). The body’s own immune system attacks the joints, causing swelling, pain, heat, redness, immobility, and tissue damage that can cause deformation (Figure 1). Because rheumatoid arthritis is an autoimmune disease, some types of immunomodulatory agents are specifically useful for treating this condition. Although both types of arthritis are associated with joint inflammation, most of the immunomodulatory drugs used for these two joint diseases are different. The exception is the non-steroidal anti-inflammatory drugs (NSAIDs) and corticosteroids.
NSAIDs decrease the production of prostaglandins by inhibiting an enzyme (cyclooxygenase) necessary for the production of these inflammatory molecules. There are several types of prostaglandins and these lipid-derived molecules have multiple actions and affect many different kinds of cells, including neurons, cells in the blood vessels (endothelial cells), and immune cells. Because NSAIDs block the production of all of the prostaglandins (and the lipid mediators that promote resolution of inflammation), they alter the immune response and thus are immunomodulatory. However, NSAIDs are not considered disease-modifying treatment.
Corticosteroids can be directly injected into the affected joints or delivered orally. Corticosteroids, such as dexamethasone or prednisone, mimic the body’s natural immunomodulating hormone, cortisol. Cortisol is a glucocorticoid hormone that is released in response to stress and low blood sugar. In addition to reducing inflammatory immune responses, drugs that mimic this hormone have many side effects, including altering the distribution of fat tissue, triggering a type of diabetes, inhibition of bone formation, and suppression of the adrenal gland. Although corticosteroids reduce inflammation and can reduce disease progression, they are typically considered as a separate category from disease-modifying therapies.

In the treatment of rheumatoid arthritis, the use of “disease-modifying antirheumatic drugs” (DMARDs) is considered critical to preserving joint tissue and limiting disease progression (3). There are two main groups of DMARDs, those that can be taken orally and those that must be injected. The oral or conventional DMARDs are chemically diverse, but all are “small-molecule” drugs. The other type of DMARDs are “biologic” drugs. Small-molecule therapies are chemicals with a defined structure that usually can be synthesized in a lab or isolated and purified to homogeneity. In contrast, biologics are usually large and complex and may have many different components (4) (Figure 2). Blood, like that used in transfusions, is a biologic. Cell-based therapies and protein-based therapies are other types of biologics. All of the DMARDs are immunomodulatory, yet they all have different mechanisms of action.
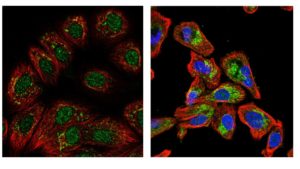
Here, the focus is the oral or conventional DMARDs. These medicines, along with corticosteroids, tend to be the first options for disease-modifying treatments for rheumatoid arthritis. They have a long history of use in patients, not just for arthritis, and the adverse effects are well known and can be effectively monitored. These treatments include methotrexate (an inhibitor of dihydrofolate reductase), sulfasalazine (mechanism of action unknown), leflunomide (an inhibitor of dihydroorotate dehydrogenase), and hydroxychloroquine (an inhibitor of lysosome function). Methotrexate, leflunomide, and hydroxychloroquine are all chemicals that enter cells to affect cell function. Methotrexate inhibits an enzyme that is localized to the mitochondria and is critical for DNA synthesis; leflunomide inhibits an enzyme found in mitochondria and the nucleus that is necessary for any reactions that involve the cofactor NADP (Figure 3). Methotrexate and leflunomide are immunomodulatory, because T cells are particularly dependent on the metabolic pathways that these drugs block.
Hydroxychloroquine has several targets. It increases the pH of intracellular compartments called lysosomes, which impairs the processing of antigens, thereby preventing antigen-induced activation of the T cells. By making lysosomes less acidic, hydroxychloroquine may also impair cytotoxicity of T cells by reducing the activity of lysosomal enzymes, which are released as part of the cytotoxic response. Hydroxychloroquine also impairs the activity of a subset of the class of immune receptors called Toll-like receptors (TLRs). Hydroxchloroquine specifically antagonizes the activity of TLRs that function in low pH parts of the cell, called endosomes. Thus, all of the immunomodulatory actions of hydroxychloroquine may relate to its ability to increase the pH of specific cellular compartments. Signaling by the endosomally located TLRs promotes the release of pro-inflammatory mediators, such as tumor necrosis factor (TNF) from cells in joints; thus, reducing TLR activity limits the release of the pro-inflammatory mediators to limit immune responses that cause tissue damage.
The next commentary in this series will describe the biologic DMARDs and immunomodulatory agents that may treat osteoarthritis.
Related Resources
(1) Rheumatoid Arthritis: Rheumatoid Arthritis Support Network (accessed on 29 January 2018) https://www.rheumatoidarthritis.org/ra/ra-vs-oa/
(2) Biologics Overview. Arthritis Foundation. (accessed 29 January 2018) http://www.arthritis.org/living-with-arthritis/treatments/medication/drug-types/biologics/drug-guide-biologics.php
(3) K. E. Donahue, D. E. Jonas, R. A. Hansen, R. Roubey, B. Jonas, L. J. Lux, G. Gartlehner, E. Harden, T. Wilkins, V. Peravali, S. I. Bangdiwala, A. Yuen, P. Thieda, L. C. Morgan, K. Crotty, R. Desai, M. Van Noord, Drug Therapy for Rheumatoid Arthritis in Adults: An Update. Comparative Effectiveness Reviews, No. 55. U. S. Agency for Healthcare Research and Quality, Rockville (MD), April 2012. Executive Summary
(4) FDA Basics Webinar, June 17, 2013: Biological Products: Part 1. U. S. Food & Drug Adminstration, U. S. Department of Health and Human Services. https://www.fda.gov/AboutFDA/Transparency/Basics/ucm355978.htm
Small-Molecule Disease-Modifying Drugs
Methotrexate (access 29 January 2018) https://www.drugbank.ca/drugs/DB00563
Leflunomide (accessed 15 January 2018) https://www.drugbank.ca/drugs/DB01097
Sulfasalazine (accessed 15 January 2018) https://www.drugbank.ca/drugs/DB00795
Hydroxychloroquine (accessed 15 January 2018) https://www.drugbank.ca/drugs/DB01611
Cite as: N. R. Gough, Immunomodulators for Arthritis: Small-Molecule Disease-Modifying Agents. BioSerendipity (29 January 2018). https://www.bioserendipity.com/immunomodulators-for-arthritis-small-molecule-disease-modifying-agents/